What is Leukocyte Adhesion Deficiency (LAD)?
Leukocyte adhesions deficiency (LAD) syndromes are a group of rare disorders affecting the immune system. LAD syndromes are characterized by defects affecting how white blood cells (leukocytes) respond and travel to the site of a wound or infection.
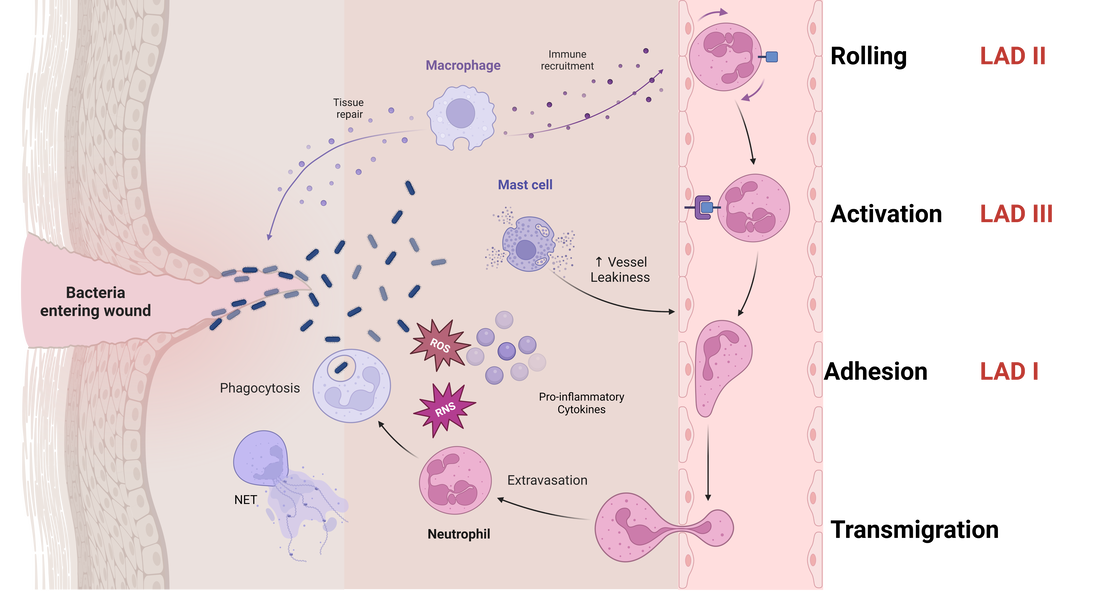
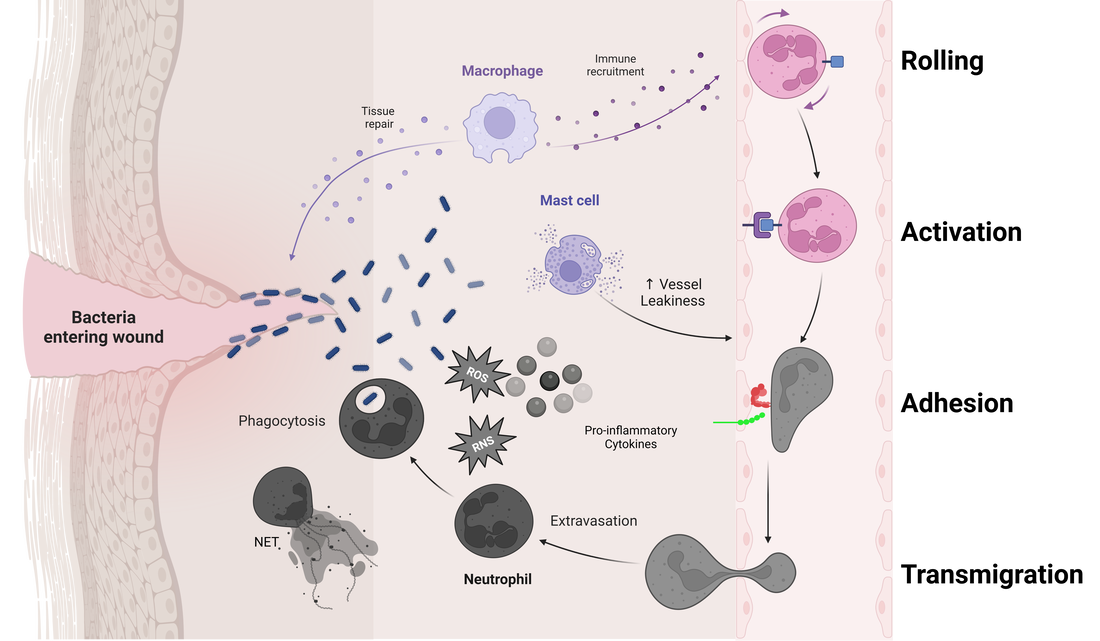
The transmigration of white blood cells to the site of inflammation is a crucial part of the body's defense against harmful pathogens. This process is highly dynamic and includes multiple stages, known as the adhesion cascade, which is facilitated by various groups of adhesion molecules. These molecules are found on both resting and stimulated endothelial cells and white blood cells. Defects in some of these adhesion molecules can lead to primary immunodeficiencies, which are collectively referred to as leukocyte adhesion deficiency (LAD) syndromes. Currently, there are three recognized syndromes, each affecting a distinct stage of the adhesion cascade.[1]
The transmigration of white blood cells to the site of inflammation is a crucial part of the body's defense against harmful pathogens. This process is highly dynamic and includes multiple stages, known as the adhesion cascade, which is facilitated by various groups of adhesion molecules. These molecules are found on both resting and stimulated endothelial cells and white blood cells. Defects in some of these adhesion molecules can lead to primary immunodeficiencies, which are collectively referred to as leukocyte adhesion deficiency (LAD) syndromes. Currently, there are three recognized syndromes, each affecting a distinct stage of the adhesion cascade.[1]
Leukocyte Adhesion Deficiency (LAD) Type 1
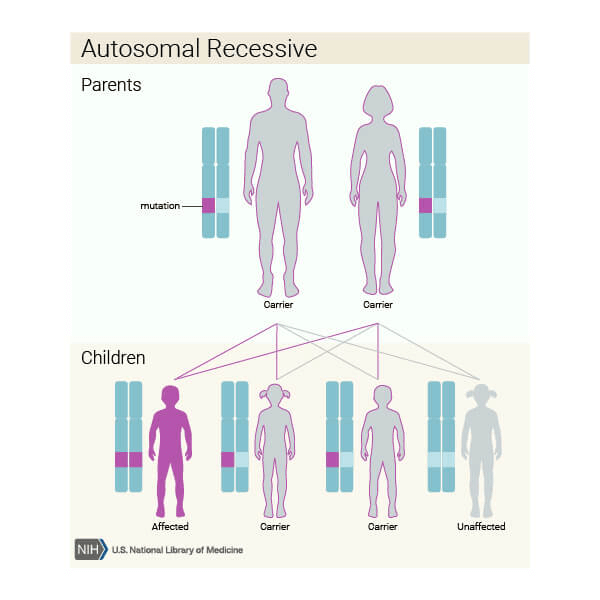
LAD I was first described almost forty years ago, it was one of the first immunodeficiency syndromes where genetic defect was found. LAD is transmitted in a autosomal recessive manner.
Prominent clinical features of LAD I includes recurrent pyogenic infections in the first weeks of life(93.3%), delayed separation of umbilical cord, severe gingivitis and periodontitis, and absence of pus formation at sites of infection. Infections are mainly caused by gram-negative bacteria like Staphylococcus aureus and Pseudomonas aeruginos. Patients with LAD I exhibit mild to moderate neutrophilia in absence of overt infection. [3]
Prominent clinical features of LAD I includes recurrent pyogenic infections in the first weeks of life(93.3%), delayed separation of umbilical cord, severe gingivitis and periodontitis, and absence of pus formation at sites of infection. Infections are mainly caused by gram-negative bacteria like Staphylococcus aureus and Pseudomonas aeruginos. Patients with LAD I exhibit mild to moderate neutrophilia in absence of overt infection. [3]
What are the phases in a adhesion cascade?
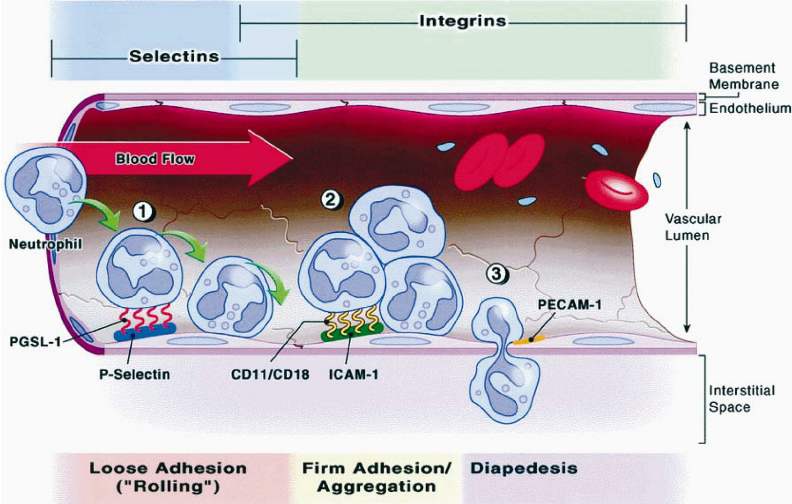
Leukocyte movement from bloodstream to issues involves four crucial phases.
1. Rolling: While the leukocytes are traveling through the vessels, loose adhesion us formed to the vessel wall, causing the leukocyte to roll on the endothelium. This step is facilitated by selectins, expressed on the activated endothelium of the vessels and saylated ligands expressed on leukocytes. This step is transient and reversible.
2. Activation: During this phase, leukocytes comes into contact with various chemokines and cytokines that will transform the inactive integrins on leukocytes into active integrins in preparation for the next phase.
3. Adhesion: This step transforms the loose adhesion between leukocyte and endothelial wall into a stronger one by bind the active integrins to their ligands- Intracellular adhesion molecules(ICAMs) on the endothelial wall.
4. Transmigration: This phase is mediated by several chemoattractive factors that will loosen the endothelial wall and guide leukocytes to the site of inflammation.
1. Rolling: While the leukocytes are traveling through the vessels, loose adhesion us formed to the vessel wall, causing the leukocyte to roll on the endothelium. This step is facilitated by selectins, expressed on the activated endothelium of the vessels and saylated ligands expressed on leukocytes. This step is transient and reversible.
2. Activation: During this phase, leukocytes comes into contact with various chemokines and cytokines that will transform the inactive integrins on leukocytes into active integrins in preparation for the next phase.
3. Adhesion: This step transforms the loose adhesion between leukocyte and endothelial wall into a stronger one by bind the active integrins to their ligands- Intracellular adhesion molecules(ICAMs) on the endothelial wall.
4. Transmigration: This phase is mediated by several chemoattractive factors that will loosen the endothelial wall and guide leukocytes to the site of inflammation.
The four phases involves a variety of signaling molecules, adhesion molecules, ligands and integrins. Defects in any of these phases will lead to Leukocyte Adhesion Deficiency disorders. Defects in the first three phases are reported in humans and animals. LAD I affects several hundreds of patients around the world, due to mutation in β2 integrin, which is essential for firm adhesion . LAD II affects less than ten patients in the world, faulted the absence of the selectin fucosylated ligands crucial for the rolling phase. LAD III is reported to affect twenty patients in the world, caused by mutations in kindlin-3, leading to abnormal integrin activation during the activation phase.[1]
Role of ITGB2 in LAD Type 1The ITGB2 gene provides instructions for making Integrin subunit β2. β2 integrins combine multiple different alpha (α) subunits to form different integrin heterodimers. Integrins play a crucial role in cell adhesion and cell-surface mediated signaling.[1]
These integrins interact with adhesion molecules on the surface of endothelial cells, such as intercellular adhesion molecule-1 (ICAM-1), to mediate firm adhesion and transmigration. The βI domain of ITGB2 plays a crucial role in ligand binding and is responsible for coordinating the interaction with ICAM-1 and other adhesion molecules. In LAD-I patients, mutations in ITGB2 lead to abnormalities in the βI domain or the overall structure of the protein, resulting in reduced affinity for ICAM-1 and impaired leukocyte adhesion. Consequently, the leukocyte transmigration cascade is disrupted, preventing leukocytes from reaching the site of infection or inflammation. This inability to initiate an effective immune response leaves LAD-I patients susceptible to recurrent bacterial and fungal infections and severely compromises their immune system. [3] Mutations in ITGB2 gene affect the level of expression in β2 integrins on the leukocyte membrane. Ranging from reduced expression to complete absence of β2 integrins. The expression variability is associated with disease severity. At least 91 mutations in the ITGB2 gene have been found to cause leukocyte adhesion deficiency type one. [2] |
|
Diagnosis and Treatment
|
|
The immunological investigation involves various laboratory tests, including immunodeficiency screening, genetic testing, complement testing, and flow cytometric analysis. LAD can be differentiated from other conditions such as sepsis and neutropenic disorders[4]. In the affected individuals, 75% of them will have the severe form of LAD, with less than 1% of ITGB2 expression, they usually succumb to life-threatening infections before one year of age. The other 25% of affected individuals have a milder form of LAD, with 1-30% of ITGB2 expression, most of them will live to adulthood with appropriate antimicrobial therapy.
Treatment for LAD -I include hematopoietic steam cell transplant (HSCT), recombinant human interferon-gamma treatment, and prophylactic immunoglobulin therapy have been proven successful. [3] |
What are the symptoms of Leukocyte Adhesion Deficiency Type - I?
Leukocyte Adhesion Deficiency Type I (LAD-I) is a rare genetic disorder that affects the immune system. People with LAD-I often experience recurrent infections, as their immune cells struggle to reach and combat infections effectively. Some common symptoms of LAD-I include:
- Frequent infections: Individuals with LAD-I are more susceptible to bacterial and fungal infections, which can affect the skin, respiratory system, and gastrointestinal tract. These infections often recur and can be difficult to treat.
- Delayed wound healing: Due to impaired immune cell function, wounds may take longer to heal in LAD-I patients. This slow healing process can result in the formation of non-healing ulcers or scars.
- Absence of pus formation: While pus formation is a typical response to infection, LAD-I patients may not exhibit this symptom. Their immune cells are unable to reach the site of infection, resulting in a lack of pus despite the presence of an infection.
- Impaired inflammatory response: People with LAD-I might not exhibit the usual signs of inflammation, such as redness, swelling, and warmth, at the site of an infection or injury. This is due to the inability of their immune cells to migrate to the affected area.
- Failure to thrive: Children with LAD-I may experience growth delays or developmental problems due to the frequent infections and the body's constant battle to overcome them.
How is it inherited?The ITGB2 gene provides instructions for making Integrin subunit β2. β2 integrins combine multiple different alpha (α) subunits to form different integrin heterodimers. Integrins play a crucial role in cell adhesion and cell-surface mediated signaling.[1]
Mutations in ITGB2 gene affect the level of expression in β2 integrins on the leukocyte membrane. Ranging from reduced expression to complete absence of β2 integrins. The expression variability is associated with disease severity. At least 91 mutations in the ITGB2 gene have been found to cause leukocyte adhesion deficiency type one. [2] |
Reference
[1] Hanna, S., & Etzioni, A. (2012, January 25). 2012 - The New York Academy of Sciences - Wiley Online Library. Leukocyte adhesion deficiencies. Retrieved February 20, 2023, from https://nyaspubs.onlinelibrary.wiley.com/doi/10.1111/j.1749-6632.2011.06389.x
[2] Roos D, van Leeuwen K, Madkaikar M, Kambli PM, Gupta M, Mathews V, Rawat A, Kuhns DB, Holland SM, de Boer M, Kanegane H, Parvaneh N, Lorenz M, Schwarz K, Klein C, Sherkat R, Jafari M, Wolach B, den Dunnen JT, Kuijpers TW, Köker MY. Hematologically important mutations: Leukocyte adhesion deficiency (second update). Blood Cells Mol Dis. 2023 Mar;99:102726. doi: 10.1016/j.bcmd.2023.102726. Epub 2023 Jan 20. PMID: 36696755.
[3] Justiz Vaillant AA, Ahmad F. Leukocyte Adhesion Deficiency. [Updated 2022 Jul 8]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK539770/
[4] Wittwer C, ARUP consult. “Leukocyte Adhesion Deficiency | Choose the Right Test.” Accessed April 11, 2023. https://arupconsult.com/content/leukocyte-adhesion-deficiency.
Images:
Header: "Blood vessels, transmitted light brightfield" https://www.zeiss.com/microscopy/en/products/light-microscopes/widefield-microscopes/axiolab-5.html
Leukocyte transmigration cascade: Created with Biorender
ITGB2 conformations slide show: Me
Sequence of events in emigration of neutrophils: https://www.researchgate.net/profile/Alexander-Seifalian/publication/8235543/figure/fig1/AS:277907470602240@1443269712236/Sequence-of-events-in-emigration-of-neutrophils-First-the-endothelial-selectins-are.png
Trigger Warning: Verde Magazine https://verdemagazine.com/trigger-warnings-self-regulation-on-media-offers-support
LAD symptoms photos:
Sharma, A., Jindal, A.K., Pilania, R.K. et al. Palatal Ulcer in Leukocyte Adhesion Deficiency: an Unusual Occurrence. J Clin Immunol 38, 736 (2018). https://doi.org/10.1007/s10875-018-0545-4
AlisonHaynes, AndrewO’Keefe, PaulDancey, KamalOhson, LesleyTurner, and MarisaChard. 2018. Leukocyte adhesion deficiency-I caused by a novel mutation in ITGB2 presenting with pyoderma gangrenosum. LymphoSign Journal. 5(3): 86-90. https://doi.org/10.14785/lymphosign-2018-0006
Fekadu, Julia, Ute Modlich, Peter Bader, and Shahrzad Bakhtiar. 2022. "Understanding the Role of LFA-1 in Leukocyte Adhesion Deficiency Type I (LAD I): Moving towards Inflammation?" International Journal of Molecular Sciences 23, no. 7: 3578. https://doi.org/10.3390/ijms23073578
Inheritance pattern: https://www.proteinatlas.org/ENSG00000160255-ITGB2/structure
[2] Roos D, van Leeuwen K, Madkaikar M, Kambli PM, Gupta M, Mathews V, Rawat A, Kuhns DB, Holland SM, de Boer M, Kanegane H, Parvaneh N, Lorenz M, Schwarz K, Klein C, Sherkat R, Jafari M, Wolach B, den Dunnen JT, Kuijpers TW, Köker MY. Hematologically important mutations: Leukocyte adhesion deficiency (second update). Blood Cells Mol Dis. 2023 Mar;99:102726. doi: 10.1016/j.bcmd.2023.102726. Epub 2023 Jan 20. PMID: 36696755.
[3] Justiz Vaillant AA, Ahmad F. Leukocyte Adhesion Deficiency. [Updated 2022 Jul 8]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK539770/
[4] Wittwer C, ARUP consult. “Leukocyte Adhesion Deficiency | Choose the Right Test.” Accessed April 11, 2023. https://arupconsult.com/content/leukocyte-adhesion-deficiency.
Images:
Header: "Blood vessels, transmitted light brightfield" https://www.zeiss.com/microscopy/en/products/light-microscopes/widefield-microscopes/axiolab-5.html
Leukocyte transmigration cascade: Created with Biorender
ITGB2 conformations slide show: Me
Sequence of events in emigration of neutrophils: https://www.researchgate.net/profile/Alexander-Seifalian/publication/8235543/figure/fig1/AS:277907470602240@1443269712236/Sequence-of-events-in-emigration-of-neutrophils-First-the-endothelial-selectins-are.png
Trigger Warning: Verde Magazine https://verdemagazine.com/trigger-warnings-self-regulation-on-media-offers-support
LAD symptoms photos:
Sharma, A., Jindal, A.K., Pilania, R.K. et al. Palatal Ulcer in Leukocyte Adhesion Deficiency: an Unusual Occurrence. J Clin Immunol 38, 736 (2018). https://doi.org/10.1007/s10875-018-0545-4
AlisonHaynes, AndrewO’Keefe, PaulDancey, KamalOhson, LesleyTurner, and MarisaChard. 2018. Leukocyte adhesion deficiency-I caused by a novel mutation in ITGB2 presenting with pyoderma gangrenosum. LymphoSign Journal. 5(3): 86-90. https://doi.org/10.14785/lymphosign-2018-0006
Fekadu, Julia, Ute Modlich, Peter Bader, and Shahrzad Bakhtiar. 2022. "Understanding the Role of LFA-1 in Leukocyte Adhesion Deficiency Type I (LAD I): Moving towards Inflammation?" International Journal of Molecular Sciences 23, no. 7: 3578. https://doi.org/10.3390/ijms23073578
Inheritance pattern: https://www.proteinatlas.org/ENSG00000160255-ITGB2/structure